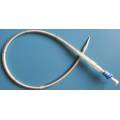
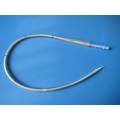
Two Stage/Disposable/Caidiac/Femoral Venous Cannula
Basic Info
Model No.: KX0206
Product Description
Model NO.: KX0206
Material: Plastic
Ethylene Oxide Sterilization: Ethylene Oxide Sterilization
Group: Adult
Size: 16fr -30fr
Specification: CE, ISO 13485, ISO 9001 GMP
HS Code: 9018390000
Type: Catheter
Certification: CE, SGS, ISO13485
Quality Guarantee Period: Two Years
Logo Printing: With Logo Printing
Trademark: Qunxing
Origin: China
Application: In cardiac operation under direct vision, it can be inserted into the femoral vein for blood flow in extracorporeal circulation.
Features:
1. Thin wall and flexible body;
2. Wirewound construction using stainless steel wire;
3. Multiple side port tip to maximize flow rate;
4. Depth marking on cannula body facilicates insertion and positioning;
5. Two stage design which more efficiently drains the heart by meeting the drainage requirements;
Model No.: KX0206
Sizes: 16FR 18FR 20FR 22FR 24FR 26FR 28FR 30FR

Contact us if you need more details on Disposable Medical. We are ready to answer your questions on packaging, logistics, certification or any Other aspects about Femoral Cannula、Cardiac Surgery Products. If these products fail to match your need, please contact us and we would like to provide relevant information.
Product Categories : Cardioplegia Circuit Cannulae
Other Products
Hot Products
Blood Collection Needle with Plastic Needle HolderNon-Woven Face MaskSetrile Disposable Insulin SyringeCE Approved Single Stage Venous CannulaAutotransfusion SystemCardiac Femoral Venous Cannula with CECurved Tip Aortic Cannula with Suture FlangeBalloon Inflation/Inflation DeviceRadial Artery Compression Device (QXZ-B)Wound Protector/Retractor (A type)Tissue Stabilizer for Heart SurgeryCE Marks Left Vents for Heart SurgeryLeft Vent Cannula with CE ApprovedPVC/Disposable/Stopcock/Aortic Root Cannula (with stopcock)Suction Tube /Medical Device /Disposable/Vent/Left Heart Sucker (KX0401)Band/Silicon/Disposable /Wound Protector - B Type