Radial Artery Compression Device (QXZ-B)
Basic Info
Model No.: kx110
Product Description
Model NO.: kx110
Material: Plastic
Ethylene Oxide Sterilization: Ethylene Oxide Sterilization
Group: Adults and Children
Size: Qxz-B
Specification: CE, ISO 13485, ISO 9001 GMP
HS Code: 9018390000
Type: Catheter
Certification: CE, SGS, ISO13485
Quality Guarantee Period: Two Years
Logo Printing: With Logo Printing
Trademark: Qunxing
Origin: Changzhou, Jiangsu
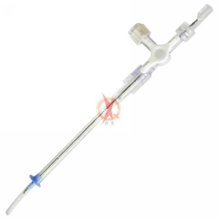
APPLICATION: This is a Compression Device to assist hemostasis of the artery after a transradial procedure.
Structure & Feature: This product is composed of a compression airbag, an air-injection port and a tie strap, as well as an appropriative inflator which can be only used to inject air and adjust the inlet and outlet air volume. It is for single use only.
Length: 240mm (or as customer's requirements)
Material: PVC & PC Contact us if you need more details on Compression Device. We are ready to answer your questions on packaging, logistics, certification or any Other aspects about Cardiology Products、Radial Artery. If these products fail to match your need, please contact us and we would like to provide relevant information.
Product Categories : Cardiology Products
Other Products
Hot Products
Blood Collection Needle with Plastic Needle HolderNon-Woven Face MaskSetrile Disposable Insulin SyringeCE Approved Single Stage Venous CannulaAutotransfusion SystemCardiac Femoral Venous Cannula with CECurved Tip Aortic Cannula with Suture FlangeBalloon Inflation/Inflation DeviceRadial Artery Compression Device (QXZ-B)Wound Protector/Retractor (A type)Tissue Stabilizer for Heart SurgeryCE Marks Left Vents for Heart SurgeryLeft Vent Cannula with CE ApprovedPVC/Disposable/Stopcock/Aortic Root Cannula (with stopcock)Suction Tube /Medical Device /Disposable/Vent/Left Heart Sucker (KX0401)Band/Silicon/Disposable /Wound Protector - B Type